The absorption of an individual electrons captured on film
The drop had a diameter of 29 micrometers, which roughly corresponds to the thickness of a thin human hair. Despite this, it contains around 3.7 x1015 negatively charged electrons.
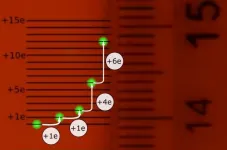
- The feat is incredible when one considers that the effect of adding single electron to a droplet that already has 3 700 000 000 000 000 is visible with the naked eye.
Now that it is possible to "see the effect of a single electron", a new opportunity emerges to better communicate science regarding elementary particles to the general public, the researchers comment.
INFORMATION:
Contact:
Javier Tello Marmolejo, doctoral student, Department of Physics, University of Gothenburg, e-mail. javier.marmolejo@physics.gu.se, phone: 070 017 53 19
Dag Hanstorp, professor, Department of Physics, University of Gothenburg, phone: 0766-22 91 41, 031-786 91 41, e-mail: dag.hanstorp@physics.gu.se
Link to article in Scientific Reports, "Visualizing the electron's quantization with a ruler" : http://www.nature.com/articles/s41598-021-89714-2
Video: Javier Marmolejo
Illustration: Javier Marmolejo
Facts about the experiment
A laser trap was used to levitate a silicone oil droplet in air. The trap consisted of a green laser with a wavelength of 532 nanometers that was directed upwards and focused by a lens with a focal distance of 100 mm. The focal point was placed between two electrodes placed in the center of the experimental chamber. The electrodes were parallel and separated by 1 mm. A 29 micrometer droplet was dropped into the laser beam, where it was trapped. Between the plates, a 666 V potential difference was applied which created a strong electric field. Alfa radiation was directed towards the almost uncharged droplet, ionizing the air around it. When the droplet gained or lost charge, the force applied by the electric field changed which in turn changed its position. The effects were magnified 73 times by a lens and projected onto a wall. With this magnification, the micrometric movements of the drop were observable with the naked eye. A common millimeter ruler was placed on the wall where the researchers could directly observe the number of electrons the drop gained as it jumped about 1 mm per added electron.