Innovative therapies for ischemic stroke: Novel bicarbonate-sensing G protein-coupled receptor shows promise
Cells actively rely on maintaining an appropriate acid-base balance to support optimal function. Under normal physiological settings, the pH inside cells remains within a controlled range. However, disruptions in this equilibrium have been linked to a wide range of health conditions, both minor and catastrophic. Changes in the extracellular environment are monitored by “membrane receptors,” of which the G protein-coupled receptors (GPCRs) are a large family of membrane proteins that mediate multiple cellular responses. However, the role of GPR30, also known as G protein-coupled estrogen receptor or membrane estrogen receptor, and its importance in cellular responses to acid-base disturbances have remained unclear.
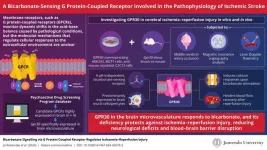
In a groundbreaking study, a team of researchers from Juntendo University in Japan, led by Specially Appointed Associate Professor Airi Jo-Watanabe investigated the cellular responses to pH variations, with a particular focus on exploring the significance of GPR30. The study was recently published in Nature Communications on 27 February 2024, with Dr. Takehiko Yokomizo, Dr. Nobutaka Hattori, and Dr. Takahiro Osada as co-authors. This study marks a crucial step in our understanding of the mechanisms governing cellular behavior in response to changes in bicarbonate concentrations. Sharing insights from the research, Dr. Jo-Watanabe explains, “Our objective was to identify a GPCR associated with the acid-base balance, and while looking for targets, GPR30 grabbed our attention. We identified GPR30 as a bicarbonate-sensing GPCR and then shifted our focus to identifying its contribution to the pathophysiology of ischemic stroke.”
So, why did the researchers choose GPR30 for their study? While screening the Psychoactive Drug Screening Program (PDSP) database, the team came across 10 GPCRs that were predominantly expressed in the stomach and pancreas, of which four were highly expressed in the brain. As pathological conditions such as ischemia and reperfusion can disrupt the acid-base balance, affecting vascular and perivascular cells through receptors, the researchers looked for GPCRs highly expressed in brain microvasculature and identified Gpr30 as one of them. This fueled curiosity about GPR30's role as a potential acid-base sensor in the brain. The team found that depleting bicarbonate from the culture medium lowered GPR30 activation (calcium responses) in GPR30-overexpressing MCF-7 and HEK cells, indicating that bicarbonate activates GPR30 in vitro.
The mouse myoblast C2C12 cell line was used to confirm that endogenous GPR30 is activated by bicarbonate ions in vitro. This was further validated ex vivo using GPR30 knock-in mice expressing the fluorescence reporter “Venus.” Confocal microscopy revealed strong expression of GPR30 in the brain microvasculature, particularly in pericytes, cells that help maintain homeostatic and hemostatic functions in the brain. This hinted at a potential mechanism for GPR30's role in cerebrovascular regulation.
Next, they decided to study the role of GPR30 in cerebral ischemia-reperfusion injury (interruption and restoration of blood flow to tissues, causing cellular dysfunction), which is key to ischemic stroke pathophysiology. GPR30 deficiency was then explored in the context of ischemia-reperfusion injury, and the researchers observed GPR30-deficient mice exhibiting significant protection against this injury, showcasing reduced neurological deficits, blood-brain barrier disruption, and apoptotic cell death. Moreover, GPR30 deficiency led to improved blood flow recovery after ischemia-reperfusion injury, emphasizing its role in controlling blood flow in both large vessels and capillaries.
The bicarbonate buffer system supplies bicarbonate ions and protons to the acid/base sensing GPCR identified in this study, modulating signal transduction in tandem with the ever-changing extracellular environment. The unexpected connection between GPR30 and bicarbonate sensing invites further exploration into the mechanisms governing cerebrovascular health, offering a potential avenue for targeted strategies to alleviate the impact of ischemic stroke reperfusion injury. Dr. Jo-Watanabe concludes by saying, “Our findings pave the way for a revolutionary approach to modulating vascular reactivity to support overall health by fine-tuning homeostatic vasoreactivity via the bicarbonate receptor.”
In conclusion, this research could mark a paradigm shift in our understanding of receptor roles in cerebrovascular regulation.
Reference
Authors
Airi Jo-Watanabe1,2, Toshiki Inaba3, Takahiro Osada4, Ryota Hashimoto5, Tomohiro Nishizawa6, Toshiaki Okuno1, Sayoko Ihara7, Kazushige Touhara7, Nobutaka Hattori3,8, Masatsugu Oh-Hora1,5, Osamu Nureki9, and Takehiko Yokomizo1
Title of original paper
Bicarbonate signalling via G protein-coupled receptor regulates ischaemia reperfusion injury
Journal
Nature Communications
DOI
10.1038/s41467-024-45579-3
Cells actively rely on maintaining an appropriate acid-base balance to support optimal function. Under normal physiological settings, the pH inside cells remains within a controlled range. However, disruptions in this equilibrium have been linked to a wide range of health conditions, both minor and catastrophic. Changes in the extracellular environment are monitored by “membrane receptors,” of which the G protein-coupled receptors (GPCRs) are a large family of membrane proteins that mediate multiple cellular responses. However, the role of GPR30, also known as G protein-coupled estrogen receptor or membrane estrogen receptor, and its importance in cellular responses to acid-base disturbances have remained unclear.
In a groundbreaking study, a team of researchers from Juntendo University in Japan, led by Specially Appointed Associate Professor Airi Jo-Watanabe investigated the cellular responses to pH variations, with a particular focus on exploring the significance of GPR30. The study was recently published in Nature Communications on 27 February 2024, with Dr. Takehiko Yokomizo, Dr. Nobutaka Hattori, and Dr. Takahiro Osada as co-authors. This study marks a crucial step in our understanding of the mechanisms governing cellular behavior in response to changes in bicarbonate concentrations. Sharing insights from the research, Dr. Jo-Watanabe explains, “Our objective was to identify a GPCR associated with the acid-base balance, and while looking for targets, GPR30 grabbed our attention. We identified GPR30 as a bicarbonate-sensing GPCR and then shifted our focus to identifying its contribution to the pathophysiology of ischemic stroke.”
So, why did the researchers choose GPR30 for their study? While screening the Psychoactive Drug Screening Program (PDSP) database, the team came across 10 GPCRs that were predominantly expressed in the stomach and pancreas, of which four were highly expressed in the brain. As pathological conditions such as ischemia and reperfusion can disrupt the acid-base balance, affecting vascular and perivascular cells through receptors, the researchers looked for GPCRs highly expressed in brain microvasculature and identified Gpr30 as one of them. This fueled curiosity about GPR30's role as a potential acid-base sensor in the brain. The team found that depleting bicarbonate from the culture medium lowered GPR30 activation (calcium responses) in GPR30-overexpressing MCF-7 and HEK cells, indicating that bicarbonate activates GPR30 in vitro.
The mouse myoblast C2C12 cell line was used to confirm that endogenous GPR30 is activated by bicarbonate ions in vitro. This was further validated ex vivo using GPR30 knock-in mice expressing the fluorescence reporter “Venus.” Confocal microscopy revealed strong expression of GPR30 in the brain microvasculature, particularly in pericytes, cells that help maintain homeostatic and hemostatic functions in the brain. This hinted at a potential mechanism for GPR30's role in cerebrovascular regulation.
Next, they decided to study the role of GPR30 in cerebral ischemia-reperfusion injury (interruption and restoration of blood flow to tissues, causing cellular dysfunction), which is key to ischemic stroke pathophysiology. GPR30 deficiency was then explored in the context of ischemia-reperfusion injury, and the researchers observed GPR30-deficient mice exhibiting significant protection against this injury, showcasing reduced neurological deficits, blood-brain barrier disruption, and apoptotic cell death. Moreover, GPR30 deficiency led to improved blood flow recovery after ischemia-reperfusion injury, emphasizing its role in controlling blood flow in both large vessels and capillaries.
The bicarbonate buffer system supplies bicarbonate ions and protons to the acid/base sensing GPCR identified in this study, modulating signal transduction in tandem with the ever-changing extracellular environment. The unexpected connection between GPR30 and bicarbonate sensing invites further exploration into the mechanisms governing cerebrovascular health, offering a potential avenue for targeted strategies to alleviate the impact of ischemic stroke reperfusion injury. Dr. Jo-Watanabe concludes by saying, “Our findings pave the way for a revolutionary approach to modulating vascular reactivity to support overall health by fine-tuning homeostatic vasoreactivity via the bicarbonate receptor.”
In conclusion, this research could mark a paradigm shift in our understanding of receptor roles in cerebrovascular regulation.
Reference
Authors
Airi Jo-Watanabe1,2, Toshiki Inaba3, Takahiro Osada4, Ryota Hashimoto5, Tomohiro Nishizawa6, Toshiaki Okuno1, Sayoko Ihara7, Kazushige Touhara7, Nobutaka Hattori3,8, Masatsugu Oh-Hora1,5, Osamu Nureki9, and Takehiko Yokomizo1
Title of original paper
Bicarbonate signalling via G protein-coupled receptor regulates ischaemia reperfusion injury
Journal
Nature Communications
DOI
10.1038/s41467-024-45579-3
END