Damaged kidneys feed gut bacteria the fuel to produce a toxin that damages kidneys further
UC Davis School of Medicine / Science
Chronic kidney disease affects roughly one in seven American adults — about 35.5 million people. Globally, the estimate reaches 788 million. For those whose kidneys fail entirely, dialysis keeps them alive by filtering waste from the blood. But one particular toxin, indoxyl sulfate, latches onto serum albumin so tightly that dialysis machines cannot pull it out. Higher levels of indoxyl sulfate correlate with worse kidney disease. And now researchers at UC Davis have mapped exactly where it comes from — and why damaged kidneys make the problem worse.
The findings, published in Science, reveal a feedback loop that begins in the gut and accelerates kidney decline in mice. The mechanism involves a common gut bacterium, a nitrogen compound the body normally handles without trouble, and an enzyme that — when blocked — appears to halt the entire destructive cycle.
Nitrate, E. coli, and indole: the chain reaction
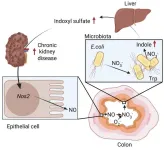
Here is how the loop works. When kidneys are impaired, they trigger increased expression of a gene called Nos2 in the colon's mucous layer. That gene produces an enzyme called inducible nitric oxide synthase (iNOS). The iNOS generates nitric oxide, which reacts with oxygen radicals to form nitrate.
Under normal conditions, nitrate in the colon is not a problem. But in the context of kidney disease, the elevated nitrate acts as rocket fuel for Escherichia coli — a common member of the Enterobacteriaceae family that normally coexists peacefully in the gut. With abundant nitrate, E. coli ramps up production of indole, an organic compound. The body converts indole into indoxyl sulfate, which circulates in the blood and inflicts further damage on already compromised kidneys.
More kidney damage means more iNOS, more nitrate, more indole, more indoxyl sulfate, and more kidney damage. The loop feeds itself.
"This study identifies nitrate from the host as a switch that turns common gut bacteria like E. coli into indole producers capable of accelerating chronic kidney disease," said Jee-Yon Lee, the study's first author.
Breaking the cycle in mice
The researchers tested whether targeting iNOS could interrupt the feedback loop. They administered aminoguanidine, an investigational drug known to inhibit iNOS, to mice with chronic kidney disease. The results were clear: treated mice showed reduced nitrate in the colonic mucus, lower indoxyl sulfate levels, and improved kidney outcomes.
Blocking a single enzyme — the one that produces nitric oxide in the gut lining — was sufficient to disrupt the entire chain. The E. coli were still present, but without elevated nitrate, they reverted to normal indole production levels. The toxin supply dropped. The kidneys stabilized.
Human gut bacteria show the same pattern
The mouse findings were not the only data point. The research team also analyzed fecal samples from people with and without chronic kidney disease. Samples from CKD patients contained higher levels of E. coli, as expected from previous research. But the critical observation was more specific: indole production from those bacteria increased only when nitrate was added to the samples. Without the nitrate trigger, the human gut bacteria behaved similarly to healthy controls.
This suggests the nitrate-dependent mechanism identified in mice also operates in the human gut — though confirming it will require clinical studies rather than laboratory assays of fecal samples.
Why this toxin is uniquely dangerous
Indoxyl sulfate presents a particular clinical problem because of its binding properties. It attaches to serum albumin — one of the most abundant proteins in blood — with enough affinity that standard hemodialysis cannot extract it. Patients on dialysis can have their blood cleaned of urea, creatinine, and other waste products, but indoxyl sulfate stays behind, continuing to damage kidney tissue and blood vessels.
This makes any strategy that reduces indoxyl sulfate production at its source — in the gut, before it reaches the bloodstream — potentially more valuable than trying to remove it after the fact.
Open questions and honest limits
The study demonstrated the mechanism in mice and showed parallel behavior in human fecal samples. But several gaps remain. Clinical trials are needed to test whether iNOS inhibitors like aminoguanidine can safely lower indoxyl sulfate and improve outcomes in people with CKD. The drug itself has not been approved for this purpose, and long-term suppression of nitrate pathways in the gut may carry trade-offs that this study did not explore.
The gut ecosystem is also more complex than any single bacterium. E. coli is not the only indole producer in the gut, and altering the nitrate environment could have downstream effects on other bacterial populations that this study did not measure.
"This study shows that altering the gut environment — not just the microbes themselves — can have profound effects on disease progression," said senior author Andreas Baumler, a distinguished professor in UC Davis's Department of Medical Microbiology and Immunology. "Targeting host pathways that shape microbial metabolism may represent a new way to intervene in chronic kidney disease."