SHP protein found to protect cartilage, offering new hope for osteoarthritis treatment
First identification of SHP protein as a key regulator that suppresses cartilage-degrading enzymes and slows osteoarthritis progression
Osteoarthritis, a condition that causes pain and reduced mobility in joints such as the knees and fingers, is one of the most common joint disorders worldwide, particularly among aging populations. The disease is characterized by the gradual breakdown of cartilage, which normally cushions the bones within joints.
Despite its prevalence, current treatments for osteoarthritis mainly focus on alleviating pain rather than addressing the underlying cause of cartilage degeneration. Effective therapies that can halt or reverse cartilage damage remain limited.
A joint research team led by Dr. Chul-Ho Lee and Dr. Yong-Hoon Kim at the Laboratory Animal Resource Center of the Korea Research Institute of Bioscience and Biotechnology (KRIBB), in collaboration with Prof. JinHyun Kim at Chungnam National University Hospital, has identified a key protein, SHP (NR0B2), that plays a critical protective role in cartilage and may offer a new therapeutic strategy for osteoarthritis.
The researchers first analyzed cartilage tissues from osteoarthritis patients and animal models of the disease. They found that the levels of SHP protein decreased significantly as the disease progressed, suggesting that loss of this protective factor contributes to accelerated cartilage destruction.
Further experiments showed that mice lacking SHP experienced more severe pain and faster cartilage degradation compared to normal mice. In contrast, restoring SHP levels in the joints led to reduced cartilage damage and improved joint function.
Mechanistic studies revealed that SHP protects cartilage by suppressing the production of matrix-degrading enzymes, specifically MMP-3 and MMP-13, which are known to break down cartilage tissue. The researchers demonstrated for the first time that SHP inhibits these enzymes at the signaling level by regulating the IKKβ/NF-κB pathway, thereby preserving cartilage integrity.
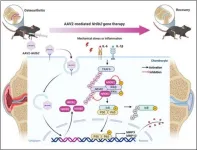
Building on these findings, the team also explored the therapeutic potential of SHP using a gene delivery approach. By injecting a viral vector carrying the SHP gene into affected joints, they observed long-lasting effects from a single treatment. Even in animals with established osteoarthritis, this approach significantly reduced cartilage damage and alleviated pain.
“This study is the first to demonstrate that the SHP protein plays a critical role in protecting cartilage during the development and progression of osteoarthritis,” said Dr. Chul-Ho Lee, the study’s lead investigator. “Therapeutic strategies targeting SHP may offer a new approach to slowing or preventing osteoarthritis progression.”
Korea Research Institute of Bioscience and Biotechnology (KRIBB) is a leading national research institute in South Korea dedicated to cutting-edge research in biotechnology and life sciences. Established in 1985, KRIBB focuses on advancing scientific knowledge in areas such as molecular biology, genomics, bioinformatics, synthetic biology, and aging-related studies. As a government-funded institute, KRIBB plays a pivotal role in driving innovation, supporting national R&D strategies, and collaborating with academic and industrial partners both domestically and internationally.
The study was published online on February 21 in Nature Communications (Impact Factor: 15.7), a leading international journal in multidisciplinary science.
The article is titled “Small heterodimer partner protects against osteoarthritis by inhibiting IKKβ/NF-κB-mediated matrix-degrading enzymes in chondrocytes.”
The corresponding authors are Dr. Chul-Ho Lee and Dr. Yong-Hoon Kim (KRIBB) and Prof. JinHyun Kim (Chungnam National University Hospital), and the first author is Dr. Eun-Jung Kang (KRIBB).
This research was supported by the Mid-career Researcher Program of the Ministry of Science and ICT and by the Major Research Programs of the Korea Research Institute of Bioscience and Biotechnology (KRIBB).
END
Despite its prevalence, current treatments for osteoarthritis mainly focus on alleviating pain rather than addressing the underlying cause of cartilage degeneration. Effective therapies that can halt or reverse cartilage damage remain limited.
A joint research team led by Dr. Chul-Ho Lee and Dr. Yong-Hoon Kim at the Laboratory Animal Resource Center of the Korea Research Institute of Bioscience and Biotechnology (KRIBB), in collaboration with Prof. JinHyun Kim at Chungnam National University Hospital, has identified a key protein, SHP (NR0B2), that plays a critical protective role in cartilage and may offer a new therapeutic strategy for osteoarthritis.
The researchers first analyzed cartilage tissues from osteoarthritis patients and animal models of the disease. They found that the levels of SHP protein decreased significantly as the disease progressed, suggesting that loss of this protective factor contributes to accelerated cartilage destruction.
Further experiments showed that mice lacking SHP experienced more severe pain and faster cartilage degradation compared to normal mice. In contrast, restoring SHP levels in the joints led to reduced cartilage damage and improved joint function.
Mechanistic studies revealed that SHP protects cartilage by suppressing the production of matrix-degrading enzymes, specifically MMP-3 and MMP-13, which are known to break down cartilage tissue. The researchers demonstrated for the first time that SHP inhibits these enzymes at the signaling level by regulating the IKKβ/NF-κB pathway, thereby preserving cartilage integrity.
Building on these findings, the team also explored the therapeutic potential of SHP using a gene delivery approach. By injecting a viral vector carrying the SHP gene into affected joints, they observed long-lasting effects from a single treatment. Even in animals with established osteoarthritis, this approach significantly reduced cartilage damage and alleviated pain.
“This study is the first to demonstrate that the SHP protein plays a critical role in protecting cartilage during the development and progression of osteoarthritis,” said Dr. Chul-Ho Lee, the study’s lead investigator. “Therapeutic strategies targeting SHP may offer a new approach to slowing or preventing osteoarthritis progression.”
Korea Research Institute of Bioscience and Biotechnology (KRIBB) is a leading national research institute in South Korea dedicated to cutting-edge research in biotechnology and life sciences. Established in 1985, KRIBB focuses on advancing scientific knowledge in areas such as molecular biology, genomics, bioinformatics, synthetic biology, and aging-related studies. As a government-funded institute, KRIBB plays a pivotal role in driving innovation, supporting national R&D strategies, and collaborating with academic and industrial partners both domestically and internationally.
The study was published online on February 21 in Nature Communications (Impact Factor: 15.7), a leading international journal in multidisciplinary science.
The article is titled “Small heterodimer partner protects against osteoarthritis by inhibiting IKKβ/NF-κB-mediated matrix-degrading enzymes in chondrocytes.”
The corresponding authors are Dr. Chul-Ho Lee and Dr. Yong-Hoon Kim (KRIBB) and Prof. JinHyun Kim (Chungnam National University Hospital), and the first author is Dr. Eun-Jung Kang (KRIBB).
This research was supported by the Mid-career Researcher Program of the Ministry of Science and ICT and by the Major Research Programs of the Korea Research Institute of Bioscience and Biotechnology (KRIBB).
END