Teaching AI to see molecular electrostatics could accelerate battery electrolyte discovery
Electrolytes sit at the heart of modern electrochemical energy storage. They control how ions move, how interfaces form, how stable a battery remains over time, and ultimately how safe and efficient the device can be. Yet discovering better electrolyte molecules remains a very challenging problem. The relevant behavior depends on subtle intermolecular interactions, solvation effects, and charge distributions, which are often expensive to resolve with quantum-chemical calculations at the scale needed for materials discovery. This is where artificial intelligence (AI) has begun to stir things up: by learning from quantum data, machine learning (ML) models can help explore chemical space much faster than conventional electronic-structure workflows.
A key quantity in this problem is the molecular electrostatic potential, or MEP. The MEP maps how a molecule feels the electrostatic attraction and repulsion in the space around it, and it is widely used to understand intermolecular interactions, molecular recognition, reactivity, and solvent design. In practical simulations, however, one rarely works directly with the full continuous electrostatic potential because obtaining it accurately is computationally demanding, calculations that can take days or even weeks.
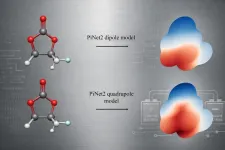
In the new study, a team of researchers from Uppsala University investigated whether ML models could infer the MEP efficiently from molecular multipole information. Using the PiNet2 architecture, the researchers trained models on dipole and quadrupole moments and tested them on the widely used QM9 dataset, a simple dataset of organic molecules ideal for studying fundamental chemical properties, as well as the broader and more complex SPICE dataset. They found that including the quadrupole moment substantially improved the ability of the model to reconstruct the electrostatic potential compared with the models that included only dipoles. The same trend was found in both datasets.
This study demonstrates that ML models trained on quadrupole moments enable rapid and accurate prediction of MEP, providing an efficient alternative to computationally intensive quantum calculations. By facilitating the characterization of electrostatic interactions in electrolyte and solvent molecules, this approach supports high-throughput screening and solvent design for energy storage devices. The results further indicate that quadrupole moments constitute a more effective training target than dipole moments for ML-based charge models, offering a practical framework for accessing electrostatic information. Overall, this methodology can accelerate the discovery and optimization of safer, more stable, and higher-performance battery solvents.
The work also highlights a broader lesson for AI in chemistry: the choice of training target matters enormously. Dipoles are often treated as the natural first target because they are the leading term in the multipole expansion of MEP for neutral molecules. But this study shows that quadrupoles carry especially valuable information when the goal is to recover the electrostatic landscape from simple point charges.
The research was carried out at Uppsala University and supported by the European Research Council, the Wallenberg Initiative Materials Science for Sustainability, and the Swedish Energy Agency.
Reference: Kadri Muuga, Lisanne Knijff, Chao Zhang. Molecular electrostatic potentials from machine learning models for dipole and quadrupole predictions[J]. AI for Science. DOI: 10.1088/3050-287X/ae531a
END
A key quantity in this problem is the molecular electrostatic potential, or MEP. The MEP maps how a molecule feels the electrostatic attraction and repulsion in the space around it, and it is widely used to understand intermolecular interactions, molecular recognition, reactivity, and solvent design. In practical simulations, however, one rarely works directly with the full continuous electrostatic potential because obtaining it accurately is computationally demanding, calculations that can take days or even weeks.
In the new study, a team of researchers from Uppsala University investigated whether ML models could infer the MEP efficiently from molecular multipole information. Using the PiNet2 architecture, the researchers trained models on dipole and quadrupole moments and tested them on the widely used QM9 dataset, a simple dataset of organic molecules ideal for studying fundamental chemical properties, as well as the broader and more complex SPICE dataset. They found that including the quadrupole moment substantially improved the ability of the model to reconstruct the electrostatic potential compared with the models that included only dipoles. The same trend was found in both datasets.
This study demonstrates that ML models trained on quadrupole moments enable rapid and accurate prediction of MEP, providing an efficient alternative to computationally intensive quantum calculations. By facilitating the characterization of electrostatic interactions in electrolyte and solvent molecules, this approach supports high-throughput screening and solvent design for energy storage devices. The results further indicate that quadrupole moments constitute a more effective training target than dipole moments for ML-based charge models, offering a practical framework for accessing electrostatic information. Overall, this methodology can accelerate the discovery and optimization of safer, more stable, and higher-performance battery solvents.
The work also highlights a broader lesson for AI in chemistry: the choice of training target matters enormously. Dipoles are often treated as the natural first target because they are the leading term in the multipole expansion of MEP for neutral molecules. But this study shows that quadrupoles carry especially valuable information when the goal is to recover the electrostatic landscape from simple point charges.
The research was carried out at Uppsala University and supported by the European Research Council, the Wallenberg Initiative Materials Science for Sustainability, and the Swedish Energy Agency.
Reference: Kadri Muuga, Lisanne Knijff, Chao Zhang. Molecular electrostatic potentials from machine learning models for dipole and quadrupole predictions[J]. AI for Science. DOI: 10.1088/3050-287X/ae531a
END