Sea creatures reveal the physics behind animal body shape diversity
New study identifies ‘mechanotypes’ as the physical links between genes and body shapes, explaining and predicting how diverse forms arise in animals like corals and sea anemones
Animals come in an extraordinary range of body shapes. A starfish looks nothing like an earthworm, a mouse, or a human. Yet even closely related species can appear radically different: corals, jellyfish, and sea anemones all belong to the same biological 'phylum', but their bodies take strikingly different forms.
A new study from EMBL researchers and their collaborators at the University of Geneva shows how such shape diversity is determined by variation in mechanical tissue properties – an idea they termed ‘mechanotypes’.
The hypothesis that physics determines biological form is not new. More than a century ago, Scottish biologist and mathematician D’Arcy Thompson published an influential tome called On Growth and Form, which emphasised the importance of physical laws in shaping the body structure of living organisms.
However, classical studies on the evolution and diversity of body shapes have focused primarily on genetics. Genotype – the genetic composition of organisms – plays a central role during growth and development. But genes alone cannot fully explain how tissues bend, stretch, and reorganise to generate body shape – a process called morphogenesis.
“Comparing genomes can reveal genetic differences linked to shape diversity, but genes cannot tell us how morphogenesis unfolds,” explained Aissam Ikmi, Group Leader at EMBL Heidelberg and one of the senior authors of the new study, recently published in the journal Cell. “Even with a genome in hand, we still cannot yet predict the final shape of an organism.”
Mapping the mechanics of morphogenesis
Ikmi and his colleagues drew on insights from mechanobiology – the study of how physical forces shape biological processes. During development, morphogenesis is often driven not by individual cells but by forces generated collectively within tissues. They hypothesised that this is the level where different body shapes arise across species.
“What matters is how cells work together as a tissue to generate forces and mechanical constraints,” explained Ikmi. “If this is where morphogenesis operates, it may also be where shape diversity emerges across evolution.”
Connecting modern biological understanding of morphogenesis to Thompson's ideas of mechanical influences on diversity required a cross-disciplinary collaboration. Ikmi Group members are experts in studying morphogenesis using animals called Cnidarians as model systems. Cnidarians – which include corals, jellyfish, and sea anemones – display a wide variety of body shapes, both in larval and adult stages, and have relatively simple body plans.
However, to build a framework that explains the physical underpinnings of this process, the study required expertise in theoretical physics and mathematics, and this was provided by Guillaume Salbreux and his postdoc Nicolas Cuny at the University of Geneva, as well as by Richard Bailleul, a mathematician by training and former EMBL postdoc.
“An important idea in physics is that when described on the right scale, emergent features of complex systems can be understood through models involving only a few key parameters,” said Salbreux.
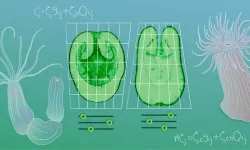
This indeed turned out to be the case for Cnidarian body shape diversity. Based on experimental observations in six different species – two corals, two anemones, and two hydrozoans – the team came up with a list of three 'mechanical modules’. These modules can be combined to explain two important features of body shape – elongation and polarity.
Elongation is a measure of how stretched or compact a body is along its main axis. Polarity, on the other hand, describes how asymmetric the animal is – whether the top part of the animal, which contains the mouth, is wider or narrower than its base. By adjusting the values of the mechanical modules in their model, like tuning knobs, scientists arrived at different predictions for elongation and polarity. They called this combination, unique for each species, an organism’s 'mechanotype’.
“Mechanical changes ultimately arise from molecular changes, but the mechanotype is where that information becomes predictive of form,” said Ikmi. “We believe evolution acts on these modules to generate new forms.”
Reshaping experiments show mechanotypes in action
Does this mean that changing the mechanotype would change the shape of the organism? To test this, the scientists performed a series of experiments using the sea anemone Nematostella. Nematostella larvae tend to be elongated and have a narrow oral end. When the scientists introduced genetic changes that affected one of the mechanical modules – nematic order – the larvae ended up being round instead of elongated. Changing polarity was more difficult though; scientists had to perturb multiple modules simultaneously to get Nematostella to change its polarity to something that resembled another species, Aiptasia.
Together, these ‘reshaping’ experiments showed it is possible to quantitatively predict and manipulate shape using mechanotypes and active surface models. They also demonstrated that different aspects of shape can be more or less complex in how they are determined by combinations of such mechanical modules.
“I think the main achievement of this study is to demonstrate the relevance of addressing morphological evolution from the perspective of mesoscopic physical principles, an idea that was proposed more than a century ago by D’Arcy Thompson and which our study now puts into practice,” said Nicolas Cuny, a postdoc in the Salbreux group and one of the first authors of the study.
A collaborative effort
The study also exemplifies how answering fundamental biological questions sometimes requires combined expertise from multiple disciplines.
“Collaboration between theorists and experimental biologists are ideal when sharing an enthusiasm for the question asked,” said Salbreux. “This was the case here: both teams were interested in the broad question of variation of shape. Besides, Aissam’s enthusiasm for figuring out principles of biology, and how experiment and theory can be used towards that goal, has been deeply inspiring.”
Ikmi agrees: “It is rare to find a collaborator you connect with both intellectually and personally; Guillaume was that for me. And this work was truly a collaborative effort: Richard and Nicolas brought expertise that shaped the analysis at every level, and the colleagues who shared Cnidarian species with us made the comparative work possible in the first place. Science like this – trying to separate signal from noise in evolution, to find the principles underneath the variation – only works when everyone is genuinely invested in the question.”
The team plans to extend their explorations of shape and investigations on the evolutionary origins of diversity, focusing next on the polyp stage in the life cycle of Cnidarians and expanding the study to include more species.
END
A new study from EMBL researchers and their collaborators at the University of Geneva shows how such shape diversity is determined by variation in mechanical tissue properties – an idea they termed ‘mechanotypes’.
The hypothesis that physics determines biological form is not new. More than a century ago, Scottish biologist and mathematician D’Arcy Thompson published an influential tome called On Growth and Form, which emphasised the importance of physical laws in shaping the body structure of living organisms.
However, classical studies on the evolution and diversity of body shapes have focused primarily on genetics. Genotype – the genetic composition of organisms – plays a central role during growth and development. But genes alone cannot fully explain how tissues bend, stretch, and reorganise to generate body shape – a process called morphogenesis.
“Comparing genomes can reveal genetic differences linked to shape diversity, but genes cannot tell us how morphogenesis unfolds,” explained Aissam Ikmi, Group Leader at EMBL Heidelberg and one of the senior authors of the new study, recently published in the journal Cell. “Even with a genome in hand, we still cannot yet predict the final shape of an organism.”
Mapping the mechanics of morphogenesis
Ikmi and his colleagues drew on insights from mechanobiology – the study of how physical forces shape biological processes. During development, morphogenesis is often driven not by individual cells but by forces generated collectively within tissues. They hypothesised that this is the level where different body shapes arise across species.
“What matters is how cells work together as a tissue to generate forces and mechanical constraints,” explained Ikmi. “If this is where morphogenesis operates, it may also be where shape diversity emerges across evolution.”
Connecting modern biological understanding of morphogenesis to Thompson's ideas of mechanical influences on diversity required a cross-disciplinary collaboration. Ikmi Group members are experts in studying morphogenesis using animals called Cnidarians as model systems. Cnidarians – which include corals, jellyfish, and sea anemones – display a wide variety of body shapes, both in larval and adult stages, and have relatively simple body plans.
However, to build a framework that explains the physical underpinnings of this process, the study required expertise in theoretical physics and mathematics, and this was provided by Guillaume Salbreux and his postdoc Nicolas Cuny at the University of Geneva, as well as by Richard Bailleul, a mathematician by training and former EMBL postdoc.
“An important idea in physics is that when described on the right scale, emergent features of complex systems can be understood through models involving only a few key parameters,” said Salbreux.
This indeed turned out to be the case for Cnidarian body shape diversity. Based on experimental observations in six different species – two corals, two anemones, and two hydrozoans – the team came up with a list of three 'mechanical modules’. These modules can be combined to explain two important features of body shape – elongation and polarity.
Elongation is a measure of how stretched or compact a body is along its main axis. Polarity, on the other hand, describes how asymmetric the animal is – whether the top part of the animal, which contains the mouth, is wider or narrower than its base. By adjusting the values of the mechanical modules in their model, like tuning knobs, scientists arrived at different predictions for elongation and polarity. They called this combination, unique for each species, an organism’s 'mechanotype’.
“Mechanical changes ultimately arise from molecular changes, but the mechanotype is where that information becomes predictive of form,” said Ikmi. “We believe evolution acts on these modules to generate new forms.”
Reshaping experiments show mechanotypes in action
Does this mean that changing the mechanotype would change the shape of the organism? To test this, the scientists performed a series of experiments using the sea anemone Nematostella. Nematostella larvae tend to be elongated and have a narrow oral end. When the scientists introduced genetic changes that affected one of the mechanical modules – nematic order – the larvae ended up being round instead of elongated. Changing polarity was more difficult though; scientists had to perturb multiple modules simultaneously to get Nematostella to change its polarity to something that resembled another species, Aiptasia.
Together, these ‘reshaping’ experiments showed it is possible to quantitatively predict and manipulate shape using mechanotypes and active surface models. They also demonstrated that different aspects of shape can be more or less complex in how they are determined by combinations of such mechanical modules.
“I think the main achievement of this study is to demonstrate the relevance of addressing morphological evolution from the perspective of mesoscopic physical principles, an idea that was proposed more than a century ago by D’Arcy Thompson and which our study now puts into practice,” said Nicolas Cuny, a postdoc in the Salbreux group and one of the first authors of the study.
A collaborative effort
The study also exemplifies how answering fundamental biological questions sometimes requires combined expertise from multiple disciplines.
“Collaboration between theorists and experimental biologists are ideal when sharing an enthusiasm for the question asked,” said Salbreux. “This was the case here: both teams were interested in the broad question of variation of shape. Besides, Aissam’s enthusiasm for figuring out principles of biology, and how experiment and theory can be used towards that goal, has been deeply inspiring.”
Ikmi agrees: “It is rare to find a collaborator you connect with both intellectually and personally; Guillaume was that for me. And this work was truly a collaborative effort: Richard and Nicolas brought expertise that shaped the analysis at every level, and the colleagues who shared Cnidarian species with us made the comparative work possible in the first place. Science like this – trying to separate signal from noise in evolution, to find the principles underneath the variation – only works when everyone is genuinely invested in the question.”
The team plans to extend their explorations of shape and investigations on the evolutionary origins of diversity, focusing next on the polyp stage in the life cycle of Cnidarians and expanding the study to include more species.
END