Cholesterol accumulation contributes to genetic movement disorder
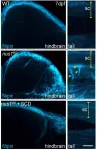
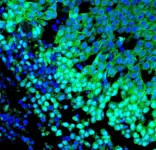
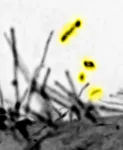
Greenwood, SC (March 22, 2021) - A research team at the Greenwood Genetic Center (GGC) has identified the mechanism that causes movement disorders in patients with mutations in the NUS1 gene. Using both cellular and model organism studies, cholesterol accumulation was found to contribute to the symptoms of seizures, ataxia, and movement abnormalities. This breakthrough study on NUS1, a gene that has also been potentially linked to Parkinson's Disease, is reported in the current issue of Genetics in Medicine, the Journal of the American College of Medical Genetics and Genomics.
Chloe Murphy, 15, of Bluffton, SC began experiencing tremors at age three. Through the years she has also experienced seizures, atypical eye movements, learning delays, ...