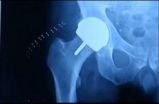
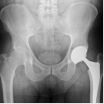
Walkup Law Office Says the DePuy ASR Hip Implant Cup Failed Long Before Expected and Advertised
After hundreds of reports of failure and loosening of the ASR cup, as well as warnings from leading physicians, and a growing number of lawsuits, DePuy finally decided to recall its defective ASR acetabular cup on August 26, 2010.
SAN FRANCISCO, CA, September 02, 2010
DePuy's ASR acetabular cup spent just over 5 years on the American market. In that time, DePuy sold over 90,000 ASR cups patients through their doctors. After hundreds of reports of failure and loosening of the ASR cup, as well as warnings from leading physicians, and a growing number of lawsuits, DePuy finally decided to recall its defective ASR acetabular cup on August 26, 2010.On the American market, the history of the DePuy ASR cup goes back to March 2004 when DePuy started a process known as "510(k)" approval. This type of FDA clearance only requires a medical device company to show that its new medical device is "substantially similar" to other devices already on the market. DePuy claimed its new ASR acetabular cup was "similar" to its prior cup, the DePuy Pinnacle device.
One crucial distinction between the Pinnacle and the ASR, however, was external fixation. While the Pinnacle was designed to be fastened to the bone with screws, the ASR offered surgeons no means to fasten it to the patient's bone. Its only means of fixation was for bone to grow into the device.
Based on the representation that the ASR was the same as a different device, DePuy began marketing and selling the ASR. DePuy and Johnson & Johnson were never required to go through extensive clinical trials or to otherwise prove that the ASR was free of dangerous defects.
When Walkup Melodia filed the first defective DePuy ASR case almost 18 months before the hip implant was recalled, our lawyers discovered that there were already more than 130 reported cases of DePuy ASR cups becoming loose and requiring revision surgery. Our first action, filed in San Francisco in March 2009, was the first defective DePuy ASR product liability case filed in the country.
Since that time, many clients have sought our experience, evaluation and representation. Our DePuy hip recall lawyers are personally prosecuting claims involving defective DePuy ASR acetabular cups requiring revision surgery on behalf of many clients.
Additional information on the DePuy ASR hip implant failures:
- DePuy ASR Hip Implant Recall Information Video (http://www.youtube.com/watch?v=pQ6BHGPzuLQ)
- Johnson & Johnson Hip Implant Recall (http://www.walkuplawoffice.com/CM/DefectiveMedicalDevices/DePuyASRHip ... ailing.asp)
- DePuy Hip Implant Recall (http://www.walkuplawoffice.com/CM/DefectiveMedicalDevices/DePuyASRHip ... awsuit.asp)
- DePuy ASR Problems Blog (http://www.walkupblog.com/2010/08/johnson-johnson-recalls-93000-depuy-asr.html)
- DePuy Hip Replacement Failure (http://www.walkuplawoffice.com/CM/WalkupNews/Walkup-Melodia-Kelly-Sch ... -Files.asp)
Patients who have received an ASR hip implant should not contact DePuy Orthopaedics or Johnson & Johnson without first discussing their legal rights with a knowledgeable and experienced attorney. You can contact us toll-free (888) 799-3968, or email us at mdavis@walkuplawoffice.com.
Website: http://www.walkuplawoffice.com