Boosting water electrolysis catalyst performance via simultaneous control of lattice distortion and oxygen vacancies!
Korea Institute of Materials Science develops high-performance non-precious metal catalyst based on iron (Fe) substitution
# A novel catalyst design enables simultaneous control of lattice structure and oxygen vacancies in molybdenum oxide through iron (Fe) substitution, with the study selected as a cover article in a leading international journal.
# Achieves high water electrolysis performance using low-cost, non-precious metal-based catalysts, with strong potential for applications in eco-friendly hydrogen production and the hydrogen economy.
CHANGWON, South Korea — Korea Institute of Materials Science (KIMS), led by President Chuljin Choi, announced that a research team led by Dr. Dahee Park at the Hydrogen Energy Materials Research Center has successfully developed a high-performance catalyst that significantly enhances the oxygen evolution reaction (OER), a key process in alkaline water electrolysis. The team achieved this by partially substituting molybdenum oxide (MoOx) with iron (Fe), enabling simultaneous control of lattice structure and oxygen vacancies. The study presents a new catalyst design strategy that delivers performance and stability comparable to precious metal catalysts while utilizing low-cost materials.
Hydrogen is widely regarded as a clean energy source with zero carbon emissions, and water electrolysis is considered a next-generation technology for eco-friendly hydrogen production. However, the oxygen evolution reaction (OER) remains a major bottleneck due to its slow kinetics and high energy requirements, which reduce overall efficiency. Although precious metal catalysts offer high performance, their high cost and limited availability have driven the need for alternative non-precious metal catalysts. Molybdenum-based oxides have attracted attention as promising alternatives due to their ability to finely tune electronic properties. However, their relatively low electrical conductivity and limited number of active sites have restricted their practical performance.
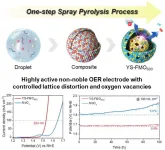
To address these challenges, the KIMS research team introduced a novel design approach by incorporating iron (Fe) into the MoOx structure, enabling simultaneous control of atomic arrangement and oxygen vacancies. This approach improves electron transport and increases the number of active sites where reactions occur. Using an aerosol-assisted spray pyrolysis process, the team successfully synthesized Fe-substituted MoOx catalysts through a single-step process. The formation of Fe–O–Mo heterostructures enhances structural stability, allowing the catalyst to maintain performance over extended operation.
Furthermore, by precisely controlling heat-treatment conditions, the researchers engineered lattice distortion and oxygen vacancies within the catalyst, forming unique core–shell and yolk–shell structures with internal voids. These structures increase the surface area in contact with water and improve electrical conductivity. Notably, the activation of the lattice oxygen mechanism (LOM), in which lattice oxygen directly participates in the reaction, was found to significantly enhance OER efficiency. As a result, the catalyst demonstrated outstanding performance, achieving a low overpotential of approximately 294 mV at a high current density of 100 mA/cm² and maintaining stable operation for over 100 hours.
This technology has strong potential for commercialization as a key catalyst for eco-friendly hydrogen production in the carbon-neutral era. In particular, it is expected to play a critical role in improving the efficiency of large-scale hydrogen production when applied to alkaline water electrolysis systems. By offering a viable alternative to expensive precious metal catalysts, the technology is also anticipated to reduce hydrogen production costs and contribute to the expansion of clean energy infrastructure.
“This study demonstrates a strategy to maximize catalytic performance by simultaneously controlling atomic structure and defects in low-cost metals,” said Dr. Dahee Park, the senior researcher at KIMS. “We plan to extend this catalyst design approach to various electrochemical energy conversion reactions and further develop next-generation eco-friendly energy technologies.”
The research was supported by the National Research Foundation of Korea under the Ministry of Science and ICT and by the Ministry of Trade, Industry and Energy. The findings were published online on February 12, 2026, in the international journal ChemSusChem (Impact Factor: 6.6) and selected as a cover article for its March 2026 issue.
-----------------------------------------------------------------------
###
About Korea Institute of Materials Science(KIMS)
KIMS is a non-profit government-funded research institute under the Ministry of Science and ICT of the Republic of Korea. As the only institute specializing in comprehensive materials technologies in Korea, KIMS has contributed to Korean industry by carrying out a wide range of activities related to materials science including R&D, inspection, testing&evaluation, and technology support.
END
# Achieves high water electrolysis performance using low-cost, non-precious metal-based catalysts, with strong potential for applications in eco-friendly hydrogen production and the hydrogen economy.
CHANGWON, South Korea — Korea Institute of Materials Science (KIMS), led by President Chuljin Choi, announced that a research team led by Dr. Dahee Park at the Hydrogen Energy Materials Research Center has successfully developed a high-performance catalyst that significantly enhances the oxygen evolution reaction (OER), a key process in alkaline water electrolysis. The team achieved this by partially substituting molybdenum oxide (MoOx) with iron (Fe), enabling simultaneous control of lattice structure and oxygen vacancies. The study presents a new catalyst design strategy that delivers performance and stability comparable to precious metal catalysts while utilizing low-cost materials.
Hydrogen is widely regarded as a clean energy source with zero carbon emissions, and water electrolysis is considered a next-generation technology for eco-friendly hydrogen production. However, the oxygen evolution reaction (OER) remains a major bottleneck due to its slow kinetics and high energy requirements, which reduce overall efficiency. Although precious metal catalysts offer high performance, their high cost and limited availability have driven the need for alternative non-precious metal catalysts. Molybdenum-based oxides have attracted attention as promising alternatives due to their ability to finely tune electronic properties. However, their relatively low electrical conductivity and limited number of active sites have restricted their practical performance.
To address these challenges, the KIMS research team introduced a novel design approach by incorporating iron (Fe) into the MoOx structure, enabling simultaneous control of atomic arrangement and oxygen vacancies. This approach improves electron transport and increases the number of active sites where reactions occur. Using an aerosol-assisted spray pyrolysis process, the team successfully synthesized Fe-substituted MoOx catalysts through a single-step process. The formation of Fe–O–Mo heterostructures enhances structural stability, allowing the catalyst to maintain performance over extended operation.
Furthermore, by precisely controlling heat-treatment conditions, the researchers engineered lattice distortion and oxygen vacancies within the catalyst, forming unique core–shell and yolk–shell structures with internal voids. These structures increase the surface area in contact with water and improve electrical conductivity. Notably, the activation of the lattice oxygen mechanism (LOM), in which lattice oxygen directly participates in the reaction, was found to significantly enhance OER efficiency. As a result, the catalyst demonstrated outstanding performance, achieving a low overpotential of approximately 294 mV at a high current density of 100 mA/cm² and maintaining stable operation for over 100 hours.
This technology has strong potential for commercialization as a key catalyst for eco-friendly hydrogen production in the carbon-neutral era. In particular, it is expected to play a critical role in improving the efficiency of large-scale hydrogen production when applied to alkaline water electrolysis systems. By offering a viable alternative to expensive precious metal catalysts, the technology is also anticipated to reduce hydrogen production costs and contribute to the expansion of clean energy infrastructure.
“This study demonstrates a strategy to maximize catalytic performance by simultaneously controlling atomic structure and defects in low-cost metals,” said Dr. Dahee Park, the senior researcher at KIMS. “We plan to extend this catalyst design approach to various electrochemical energy conversion reactions and further develop next-generation eco-friendly energy technologies.”
The research was supported by the National Research Foundation of Korea under the Ministry of Science and ICT and by the Ministry of Trade, Industry and Energy. The findings were published online on February 12, 2026, in the international journal ChemSusChem (Impact Factor: 6.6) and selected as a cover article for its March 2026 issue.
-----------------------------------------------------------------------
###
About Korea Institute of Materials Science(KIMS)
KIMS is a non-profit government-funded research institute under the Ministry of Science and ICT of the Republic of Korea. As the only institute specializing in comprehensive materials technologies in Korea, KIMS has contributed to Korean industry by carrying out a wide range of activities related to materials science including R&D, inspection, testing&evaluation, and technology support.
END