New study uncovers how genes influence retinal aging and brain health
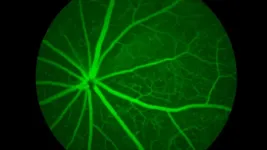
Vision changes are an inevitable part of aging, but why are some more susceptible to age-related eye diseases and why do some individuals experience more severe decline than others? New research from The Jackson Laboratory (JAX) reveals that genetics play a key role in how the eye ages, with different genetic backgrounds influencing retinal aging in distinct ways.
The study, published in Molecular Neurodegeneration, examined age-related changes in genes and proteins of the retinas of nine strains of mice, ...