Three mechanical 'knobs' predict body shape across corals, jellyfish, and anemones
Why does a coral look nothing like a jellyfish, even though they are relatives? Genetics can tell us they diverged from a common ancestor, and it can catalog the molecular differences between them. But a genome alone cannot predict what shape an organism will take. Something is missing from that equation - and a team at EMBL Heidelberg and the University of Geneva now argues that the missing variable is physics.
D'Arcy Thompson's century-old intuition, finally tested
The idea that physical forces shape biological form is older than molecular biology itself. In 1917, the Scottish biologist and mathematician D'Arcy Thompson published On Growth and Form, arguing that physical laws played a central role in determining the structure of living organisms. The book was influential but largely sidelined as genetics took over developmental biology. Genes became the dominant explanation for why organisms look the way they do.
But genes encode proteins, not shapes. The process that converts genetic instructions into three-dimensional form - morphogenesis - involves cells collectively generating forces, bending tissues, and reorganizing structures. It is inherently mechanical. Aissam Ikmi, a group leader at EMBL Heidelberg and senior author of the new study, saw an opportunity to bridge the gap.
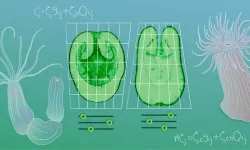
The study, published March 20 in Cell, focuses on cnidarians - the phylum that includes corals, jellyfish, and sea anemones. These animals share a relatively simple body plan but display a striking variety of shapes, both as larvae and as adults. That combination of simplicity and diversity made them ideal test subjects for a physics-based theory of form.
Three modules that control elongation and polarity
Ikmi's team, working with physicist Guillaume Salbreux and his postdoc Nicolas Cuny at the University of Geneva, along with mathematician Richard Bailleul, a former EMBL postdoc, studied six cnidarian species: two corals, two anemones, and two hydrozoans. They measured how tissues in each species behaved mechanically during development, then distilled those observations into a model.
Three mechanical modules emerged. These modules describe how tissues generate force and resist deformation at the mesoscopic scale - the level between individual cells and the whole organism. By adjusting the values of these modules, the researchers could predict two key features of body shape: elongation (how stretched or compact the body is along its main axis) and polarity (whether the oral end, containing the mouth, is wider or narrower than the base).
Each species had a unique combination of module values. The team called this combination a mechanotype - a mechanical fingerprint that links tissue properties to body shape. Where a genotype catalogs an organism's DNA, a mechanotype catalogs the physical behaviors of its tissues that actually produce form.
Reshaping a sea anemone by tuning the knobs
Prediction is one thing. Manipulation is another. To test whether mechanotypes actually determine shape, the team performed a series of experiments on the sea anemone Nematostella.
Nematostella larvae are normally elongated with a narrow oral end. When the researchers introduced genetic changes that disrupted one mechanical module - nematic order, which describes how cells align within tissues - the larvae became round instead of elongated. The model had predicted this outcome.
Changing polarity proved harder. The team had to perturb multiple modules simultaneously to shift Nematostella's polarity toward something resembling another species, Aiptasia. This result was itself informative: it showed that different aspects of shape are controlled by different combinations of mechanical parameters, and that some shape features are more robust to perturbation than others.
The experiments demonstrated that body shape could be quantitatively predicted and deliberately altered using the mechanotype framework. Change the physics, and you change the form.
Where genes end and physics begins
The mechanotype concept does not displace genetics. Mechanical changes in tissues ultimately arise from molecular changes - different gene expression patterns lead to different proteins, which lead to different cell behaviors, which lead to different tissue mechanics. But Ikmi argues that the mechanotype is the level at which genetic variation becomes predictive of form.
Consider an analogy: knowing the chemical composition of steel tells you its molecular structure but not the shape of a bridge. The shape emerges from how the material responds to forces - its mechanical properties combined with the loads placed on it. Similarly, a genome provides the raw materials, but the mechanotype describes how those materials behave under the forces of development.
The researchers propose that evolution acts on these mechanical modules to generate new body shapes. Small changes in gene expression can shift a tissue's mechanical properties, which can alter the organism's mechanotype, which can produce a different form. This provides a concrete, testable mechanism for how morphological diversity arises - not through dramatic genetic overhauls, but through tuning a small number of physical parameters.
What the framework does not yet cover
The study has important boundaries. The six species examined are all cnidarians - animals with relatively simple, radially symmetric body plans. Whether the three-module framework scales to more complex organisms with bilateral symmetry, segmented body plans, or internal skeletons remains an open question. Vertebrate morphogenesis involves many more tissue types, mechanical interactions, and developmental stages.
The model also describes larval body shapes. Adult cnidarians undergo further morphological changes during their life cycles, and the team has not yet tested whether the mechanotype framework extends to later developmental stages. They plan to examine the polyp stage next and expand the analysis to additional species.
The experiments were conducted on a limited number of species within a single phylum. A broader comparative study - including organisms from different phyla - would be needed to determine whether mechanotypes represent a universal principle or a feature specific to animals with simple body plans.
There is also the challenge of measuring mechanical properties in living tissues. The techniques used here required specialized equipment and expertise in both experimental biology and theoretical physics. Scaling this approach to large comparative studies will demand further methodological development.
Physics and biology, reunited after a century
Cuny, one of the study's first authors, framed the work as a practical realization of Thompson's original vision. For over a hundred years, the idea that physical principles govern morphological diversity remained largely theoretical. This study puts it into practice with specific, measurable parameters that can predict and alter biological form.
The collaboration itself reflected the interdisciplinary nature of the question. Ikmi's group brought expertise in cnidarian biology and experimental morphogenesis. Salbreux and Cuny contributed theoretical physics and mathematical modeling. Bailleul provided mathematical analysis. Colleagues at other institutions shared the cnidarian species that made the comparative work possible.
The result is a framework that sits between genetics and morphology - a missing layer that may help explain how evolution generates the extraordinary diversity of animal body shapes from a relatively modest toolkit of physical mechanisms.