Iron-laced biochar destroys 90% of antibiotics in water using low-energy sound waves
Published in Biochar, Shenyang Agricultural University
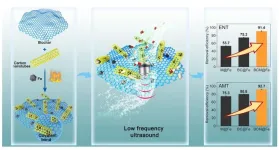
A composite material made from biochar, carbon nanotubes, and iron carbide can strip more than 90% of two common antibiotics from water when paired with low-frequency ultrasound -- achieving removal rates 15 times higher than conventional materials at a fraction of the energy cost. The findings, published in the journal Biochar, describe a system that could make treating antibiotic-contaminated wastewater significantly cheaper and more practical.
Antibiotics that water treatment plants were not designed to handle
Enrofloxacin and amoxicillin are workhorses of human and veterinary medicine. They are also persistent environmental pollutants. Wastewater treatment plants were engineered to handle organic matter and common pathogens, not synthetic pharmaceutical compounds built to resist biological degradation. The result: antibiotic residues pass through treatment systems, accumulate in rivers and groundwater, and contribute to the spread of antibiotic-resistant bacteria -- a public health crisis the World Health Organization has called one of the greatest threats to global health.
Existing removal strategies have limitations. Activated carbon adsorbs antibiotics but does not destroy them, creating a contaminated waste stream that requires further handling. Advanced oxidation processes can break down drug molecules but typically demand high energy inputs. Ultrasound-based treatment -- which uses sound waves to create microscopic bubbles that implode with enough force to generate localized temperatures above 4,000 degrees Celsius and reactive chemical species -- works in principle but has been too energy-intensive for practical deployment at low frequencies.
How the composite supercharges bubble collapse
The research team, led by Wang, Zhao, He, and colleagues, designed their material to solve that efficiency problem. The composite structure combines three components, each with a specific role.
Biochar -- carbon produced by heating biomass in low-oxygen conditions -- provides a stable, hydrophobic scaffold. Its surface texture creates nucleation sites where cavitation bubbles preferentially form and anchor. More bubbles, forming in predictable locations, means more violent collapses and more reactive chemistry per unit of sound energy.
Carbon nanotubes thread through the biochar matrix, adding mechanical strength and electrical conductivity. They also participate directly in chemical reactions, helping generate reactive oxygen species (ROS) -- highly aggressive molecular fragments like hydroxyl radicals that attack and fragment antibiotic molecules.
Iron carbide (Fe3C) nanoparticles embedded throughout the structure act as catalytic sites. Iron-based catalysts are well established in Fenton-type chemistry, where iron ions react with hydrogen peroxide to produce hydroxyl radicals. Here, the iron carbide serves a similar function, generating ROS during the extreme conditions created by bubble collapse.
The synergy matters more than any single component. Ultrasound creates the energy; the material focuses and amplifies it. The biochar surface concentrates antibiotic molecules through adsorption, bringing them into direct contact with the reactive species generated during cavitation. It is a two-stage process -- trap, then destroy -- and it runs at low ultrasound frequencies that require far less electrical power than high-frequency alternatives.
Performance numbers that stand out
In laboratory tests, the system removed more than 90% of both enrofloxacin and amoxicillin within several hours. That alone would be noteworthy. But the comparison to conventional sonocatalytic materials is where the numbers become striking: removal rates were approximately 15 times higher than those achieved by standard materials under the same conditions.
The composite also proved durable. It maintained high efficiency across a wide pH range -- important because real wastewater is chemically inconsistent -- and held up through multiple reuse cycles without significant performance loss. Tests in actual water samples, rather than idealized laboratory solutions, showed only a modest reduction in effectiveness, suggesting the material can handle the chemical complexity of real-world wastewater.
Adsorption first, degradation second
The dual mechanism is central to understanding why the system works as well as it does. Antibiotic molecules first adsorb onto the composite surface through hydrophobic interactions and molecular bonding -- essentially sticking to the material's carbon-rich surface. This concentrates the pollutants precisely where cavitation-generated reactive species are most abundant.
Then the ROS attack. Hydroxyl radicals and superoxide ions fragment the antibiotic molecules into smaller, less harmful compounds. Because the antibiotics are already immobilized on the surface rather than floating freely in solution, the probability of contact between pollutant and radical increases dramatically. The ultrasound also continuously refreshes the material surface by dispersing degradation products, preventing the kind of fouling that degrades performance in purely adsorption-based systems.
What has not been tested yet
The work was conducted at laboratory scale, and the gap between a bench-top reactor and a municipal wastewater treatment plant is vast. The study did not address several practical questions that would need answers before deployment: How does the material perform in continuous-flow systems rather than batch reactors? What is the long-term fate of iron released from the composite -- does it accumulate in treated water at concerning levels? How does the manufacturing cost compare to established treatment technologies when scaled to thousands of liters per hour?
The study also focused on two specific antibiotics. Wastewater typically contains complex mixtures of pharmaceuticals, personal care products, and industrial chemicals that may interact in unpredictable ways. Whether the composite handles mixed contamination as effectively as it handles individual compounds in clean solution is an important unanswered question.
The biochar itself is produced from biomass, but the addition of carbon nanotubes and iron carbide introduces materials with their own environmental and cost considerations. Carbon nanotube production remains energy-intensive, and the lifecycle sustainability of the composite -- from raw materials through end-of-life disposal -- deserves scrutiny.
A platform, not just a product
The researchers position their work as more than a solution for antibiotic contamination. The underlying principle -- engineering carbon materials to control where and how cavitation occurs -- could apply to other persistent organic pollutants in water, from pesticides to industrial dyes. If the approach generalizes, it represents a design strategy for a class of water treatment materials, not just a single application.
For now, the data from this study establish that the biochar-nanotube-iron carbide composite achieves antibiotic removal rates that conventional materials cannot match, at energy inputs that conventional ultrasound treatment cannot achieve. Whether those laboratory results survive contact with the messy realities of industrial wastewater treatment is the question that field-scale testing will need to answer.