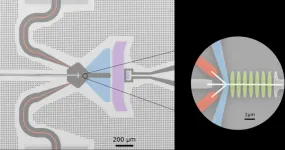
Insilico Medicine receives IND approval for novel AI-designed USP1 inhibitor for cancer
Insilico Medicine (“Insilico”), a generative artificial intelligence (AI)-driven clinical stage drug discovery company, today announced that the U.S. Food and Drug Administration (FDA) recently approved the initial investigational new drug (IND) application for ISM3091 for the treatment of patients with solid tumors. This is Insilico's first oncology program to advance to the clinical validation stage.
The open-label, multicenter Phase I clinical trial of ISM3091 will be conducted simultaneously in the U.S. and China ...