A single protein keeps cartilage-destroying enzymes in check - and gene therapy restores it
A protein called SHP (also known as NR0B2) acts as a natural shield for joint cartilage, and its levels plummet as osteoarthritis progresses. Restoring it through a single gene therapy injection reduced cartilage destruction and relieved pain in mice with established disease. The findings, published February 21 in Nature Communications, identify the first direct protective role for SHP in cartilage and suggest a new therapeutic angle for a condition that currently has no disease-modifying treatment.
The cartilage protection gap
Osteoarthritis affects hundreds of millions of people worldwide. It is the most common joint disorder in aging populations, and its hallmark is the gradual destruction of cartilage - the smooth, slippery tissue that cushions the ends of bones where they meet at joints. Once that cartilage erodes, bone grinds against bone, producing pain, stiffness, and progressive loss of mobility.
The treatments available today manage symptoms. Anti-inflammatory drugs reduce pain. Physical therapy preserves mobility. Joint replacement surgery is effective but invasive and typically reserved for severe cases. What does not exist is a drug that slows or halts the cartilage destruction itself. Every therapy on the market addresses the consequences of osteoarthritis, not its mechanism.
That gap persists partly because the molecular machinery driving cartilage breakdown is complex and redundant. Multiple enzymes, signaling pathways, and inflammatory mediators contribute, and blocking any single one has not been enough to produce lasting clinical benefit. The new study from Korea's Research Institute of Bioscience and Biotechnology (KRIBB) takes a different approach: instead of blocking a destructive pathway, it restores a protective one.
SHP disappears as joints deteriorate
The research team, led by Dr. Chul-Ho Lee and Dr. Yong-Hoon Kim at KRIBB in collaboration with Prof. JinHyun Kim at Chungnam National University Hospital, began by examining cartilage tissue from osteoarthritis patients and from animal models of the disease. They measured levels of SHP - a nuclear receptor protein previously studied in liver metabolism but not well characterized in joints.
The pattern was consistent: as osteoarthritis progressed, SHP levels in cartilage dropped. The more advanced the disease, the less SHP remained. This correlation raised an obvious question: is SHP loss a consequence of cartilage damage, or does it contribute to the damage?
Mice without SHP develop worse arthritis
To answer that, the researchers turned to mice genetically engineered to lack SHP entirely. These knockout mice developed more severe joint damage and showed greater pain responses compared to normal mice when osteoarthritis was induced. The absence of SHP did not merely correlate with worse outcomes - it produced them.
The reverse experiment confirmed the relationship. When the team restored SHP levels in affected joints, cartilage damage decreased and joint function improved. SHP was not just a bystander; it was actively protecting cartilage.
How SHP blocks the wrecking crew
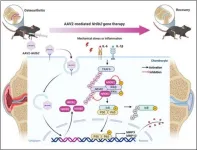
The mechanistic work revealed how. Cartilage destruction in osteoarthritis is largely carried out by matrix metalloproteinases - enzymes designated MMP-3 and MMP-13 - that chew through the collagen and proteoglycan matrix that gives cartilage its structure. These enzymes are produced by chondrocytes, the cells that maintain cartilage, when those cells receive inflammatory signals through a pathway called IKKbeta/NF-kB.
SHP, the researchers demonstrated, intervenes in this signaling chain. It suppresses IKKbeta activity, which in turn reduces NF-kB-driven production of MMP-3 and MMP-13. With SHP present, the destructive enzymes stay at low levels and cartilage remains intact. Without SHP, the brakes come off and the enzymes accumulate.
This is the first time SHP has been shown to regulate this pathway in chondrocytes. The protein was already known to play roles in bile acid metabolism and liver function, but its protective function in joints was previously unrecognized.
One injection, lasting effect
The therapeutic angle came through gene delivery. The team packaged the SHP gene into a viral vector (adeno-associated virus) and injected it directly into the affected joints of mice with established osteoarthritis. A single injection produced sustained SHP expression in the joint, and the treated mice showed significantly less cartilage damage and reduced pain compared to untreated controls.
The durability of the effect is notable. Unlike a drug that must be administered repeatedly, the gene therapy approach produced lasting protein expression from a single treatment. For a chronic, progressive disease like osteoarthritis, that kind of durability matters.
From mouse joints to human clinics - a long road
The caveats here are substantial, and they are worth stating plainly.
This is mouse research. Mice are not humans, and their joints experience different mechanical loads, have different regenerative capacities, and age on different timescales. Many therapies that work in mouse models of osteoarthritis have failed to translate to human patients. The history of osteoarthritis drug development is littered with promising preclinical results that dissolved in clinical trials.
The study used a surgically induced model of osteoarthritis, which creates a specific pattern of damage that may not fully replicate the slow, complex progression of the disease in aging human joints. The sample sizes, while adequate for demonstrating proof of concept, are small by clinical standards.
Gene therapy delivered by viral vector raises its own questions. Safety, immune responses to the vector, long-term gene expression stability, and the risk of off-target effects all need extensive testing before any human application. Joint-directed gene therapy is a concept with growing interest in the field, but no gene therapy for osteoarthritis has yet reached late-stage clinical trials.
And while SHP clearly protects against MMP-mediated cartilage destruction, osteoarthritis involves more than just enzyme activity. Inflammation, mechanical stress, subchondral bone remodeling, and synovial changes all contribute to disease progression. A therapy that addresses one axis of the disease may not be sufficient on its own.
A target worth pursuing
What the study does establish is a clear, previously unknown protective mechanism. SHP levels fall as osteoarthritis progresses, its absence accelerates damage, and restoring it reduces destruction through a well-defined molecular pathway. Whether SHP restoration becomes a clinical therapy depends on years of additional work, but the target itself - a natural cartilage-protective protein that can be delivered by gene therapy - adds something genuinely new to a field that has struggled to move beyond symptom management.