Enhanced tumor modeling using Laponite bioinks for 3D bioprinting
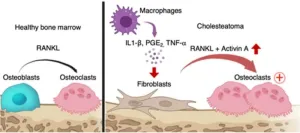
(LOS ANGELES) – August 3, 2023 - Scientists from the Terasaki Institute for Biomedical Innovation (TIBI) have developed a nanoengineered bioink with improved bonding and cross-linking capabilities for 3D bioprinting of tumor models. A key component of this bioink is Laponite, highly charged, disk-shaped, crystalline nanoparticles. As explained in their recent paper in Biofabrication, these nanoparticles were shown to enhance the biological signaling that occurs in the tumor microenvironment ...