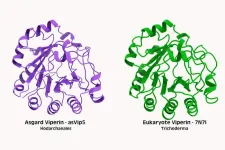
USAMMDA commercial partner receives FDA emergency use authorization for plasma powder
A U.S. Army Medical Materiel Development Activity commercial partner received Emergency Use Authorization from the U.S. Food and Drug Administration for the Department of Defense to use octaplasLG Powder—a potentially lifesaving treatment option for blood replacement therapies in certain operational circumstances. Notice of the EUA for this product was received by the company, Octapharma USA, on Aug. 8, 2024.
USAMMDA’s Warfighter Protection and Acute Care Project Management Office, which has a Cooperative Research and Development Agreement with Octapharma USA, manages research and development efforts for several ...